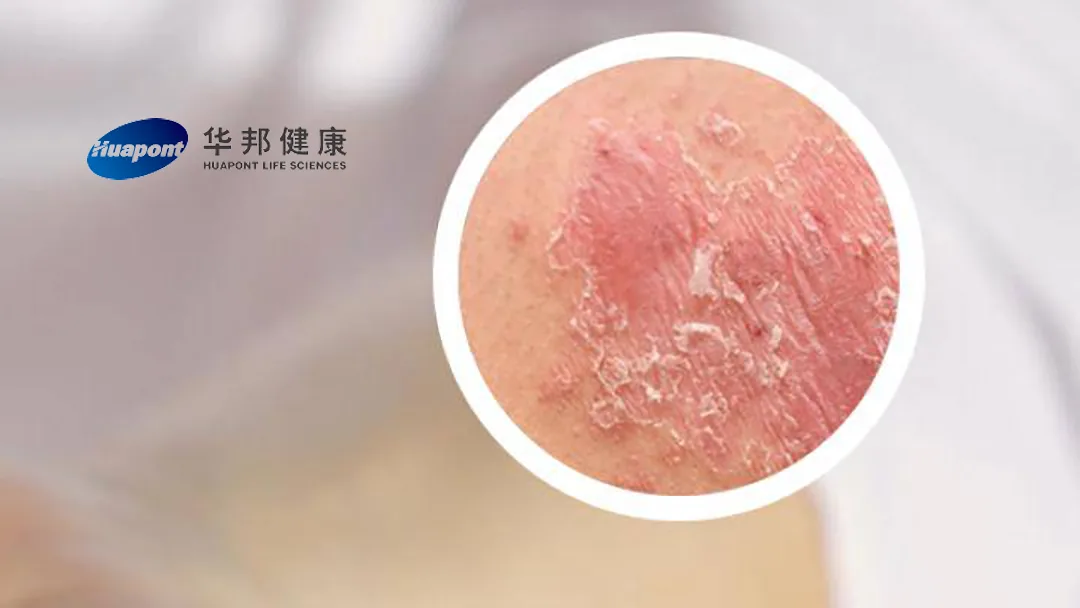
China-based Huapont Life Sciences Co., Ltd (SHE: 002004) announced that it has received marketing approval from the National Medical Products Administration (NMPA) for its generic version of LEO Pharma’s Daivobet (calcipotriol, betamethasone). This approval marks the first generic version of Daivobet to be approved in China.
Drug Composition and Mechanism
Daivobet is a compound preparation containing calcipotriol, a vitamin D analogue that regulates the proliferation and differentiation of keratinocytes, thereby reducing skin lesions, and betamethasone, which provides anti-inflammatory, anti-itch, and immunosuppressive effects to alleviate the inflammatory response associated with psoriasis.
Market Impact
With LEO Pharma’s Daivobet being the only product listed in China prior to this approval, Huapont’s generic version is now the first copycat available in the country. This development is expected to increase accessibility and provide more treatment options for patients with psoriasis.-Fineline Info & Tech