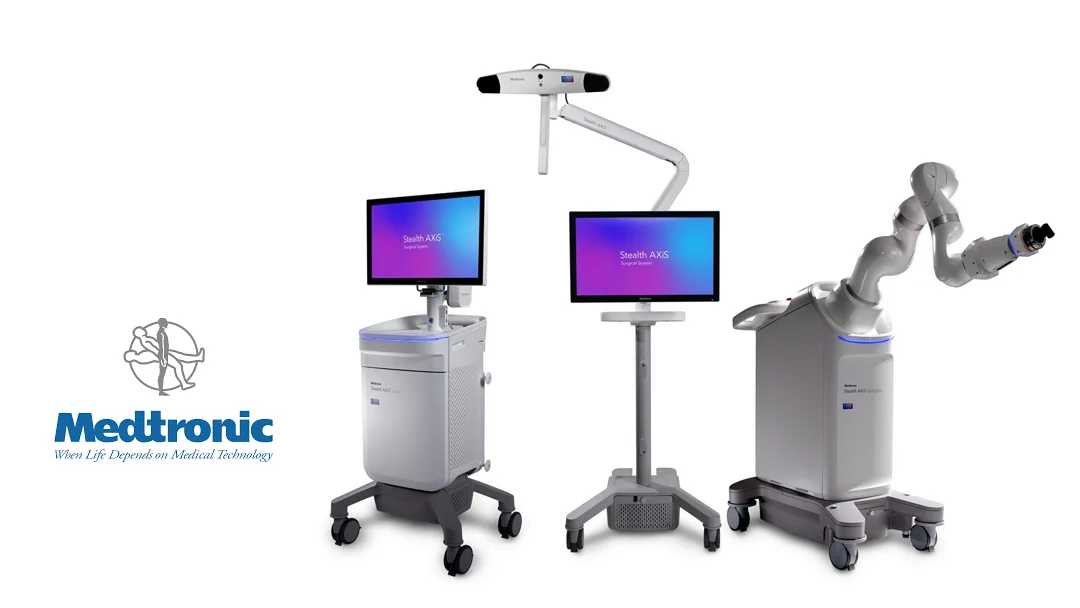
Medtronic plc (NYSE: MDT) announced it has received U.S. Food and Drug Administration (FDA) marketing approval for its Stealth AXiS™ surgical system in cranial and ear, nose, and throat (ENT) surgeries, following its initial spinal surgery clearance in February 2026. The unified platform—integrating surgical planning, real-time navigation, and robotic assistance—is designed to enhance precision, reduce procedural variability, and deliver actionable intraoperative insights during complex head and neck interventions.
Regulatory Milestones
| Indication | FDA Status | Date |
|---|---|---|
| Spinal Surgery | Cleared | February 2026 |
| Cranial & ENT Surgery | Approved | March 2026 |
| Platform Name | Stealth AXiS™ | — |
This dual-indication authorization establishes Stealth AXiS as Medtronic’s first cross-specialty intelligent surgical platform, bridging neurosurgery, ENT, and spine care under a single technological architecture.
System Capabilities
The Stealth AXiS system combines three core functions into one seamless workflow:
- Preoperative Planning: AI-assisted 3D anatomical modeling from CT/MRI scans
- Intraoperative Navigation: Sub-millimeter tracking of instruments relative to critical structures (e.g., optic nerve, carotid artery)
- Robotic Assistance: Stabilized, tremor-filtered instrument guidance with haptic feedback and safety boundaries
Unlike modular systems requiring separate consoles, Stealth AXiS operates on an integrated hardware-software ecosystem, minimizing setup time and OR footprint.
Clinical & Commercial Impact
- Procedural Precision: Enables safer access to deep-seated lesions in skull base, paranasal sinuses, and temporal bone
- Training Standardization: Reduces learning curve for complex cranial/ENT approaches through guided workflows
- Revenue Synergy: Leverages Medtronic’s existing $3B+ surgical robotics and navigation infrastructure across 4,000+ U.S. hospitals
The expansion into cranial and ENT aligns with Medtronic’s strategy to consolidate its portfolio around intelligent, data-driven surgical platforms, competing directly with offerings from Stryker, Zimmer Biomet, and Intuitive.
Market Context
- ENT Robotics Gap: Few FDA-cleared robotic systems exist for sinus or skull base surgery; most rely on manual endoscopy
- Neurosurgery Demand: Rising volumes of minimally invasive tumor resections and epilepsy procedures drive need for enhanced navigation
- Reimbursement Tailwinds: CMS and private payers increasingly support technology-enabled precision surgery with improved outcomes
With full commercial rollout expected in Q2 2026, Stealth AXiS could capture significant share in the $1.2B U.S. cranial/ENT navigation market.
Forward‑Looking Statements
This brief contains forward-looking statements regarding adoption rates, clinical outcomes, and competitive positioning. Actual results may vary due to hospital procurement cycles, surgeon training timelines, and regulatory developments in international markets.-Fineline Info & Tech