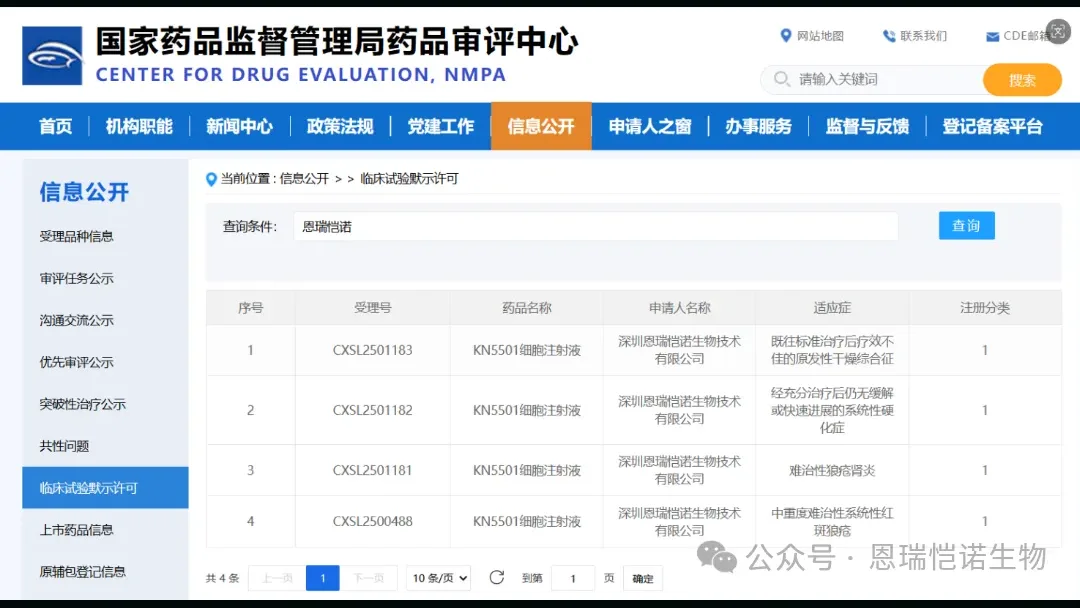
Rui Therapeutics, a China-based cell therapy developer, announced it has received approval from the National Medical Products Administration (NMPA) to initiate clinical studies for KN5501, a CD19-targeted CAR-NK therapy, in three severe autoimmune conditions: primary Sjögren’s syndrome, systemic sclerosis, and refractory lupus nephritis.
Clinical Development Plan
| Indication | Patient Population | Current Treatment Gap |
|---|---|---|
| Primary Sjögren’s Syndrome | Inadequate response to prior standard therapy | Limited effective options for refractory cases |
| Systemic Sclerosis | No remission or rapid progression after adequate treatment | High unmet need in progressive disease |
| Refractory Lupus Nephritis | Treatment-resistant kidney involvement in SLE | Significant morbidity despite current therapies |
Drug Profile & Mechanism of Action
KN5501 Characteristics
- Platform: CD19-targeted CAR-NK (Chimeric Antigen Receptor Natural Killer) cell therapy
- Target: CD19-positive B cells
- Mechanism: Precise recognition and deep elimination of pathogenic B cells
- Immunological Effect: Blocks aberrant immune responses while inducing immune system reconstitution
- Therapeutic Goal: Deep or complete remission at root cause level
Differentiated Approach
- B Cell Depletion: Comprehensive elimination of CD19+ B cells including plasma cell precursors
- Immune Reset: Promotes reconstitution of healthy immune system
- Treatment Freedom: Aims to eliminate long-term dependence on corticosteroids and immunosuppressants
- Safety Profile: NK cell platform potentially offers improved safety versus CAR-T approaches
Strategic Significance
Novel Application of Cell Therapy
- Paradigm Shift: First application of CAR-NK technology in autoimmune diseases
- Curative Potential: Moves beyond symptom management toward disease modification
- Platform Validation: Demonstrates versatility of CAR-NK platform beyond oncology
- Chinese Innovation: Represents cutting-edge cell therapy development in China
Patient Impact
- Quality of Life: Substantial improvement through elimination of chronic immunosuppression
- Side Effect Reduction: Minimizes long-term complications from corticosteroids and immunosuppressants
- Disease Control: Potential for sustained remission without continuous medication
- Economic Benefit: Reduces lifetime healthcare costs associated with chronic autoimmune management
Market Opportunity Analysis
Target Disease Populations
- Sjögren’s Syndrome: Affects 0.5-1% of general population, with significant refractory subset
- Systemic Sclerosis: Rare but severe connective tissue disease with high mortality
- Lupus Nephritis: Occurs in 40-60% of SLE patients, leading cause of morbidity/mortality
Commercial Potential
- Premium Pricing: Cell therapy pricing model supports high-value proposition
- Orphan Indications: Several target populations qualify for orphan drug incentives
- Global Applicability: Successful development supports international regulatory submissions
- Platform Expansion: Success enables expansion to additional autoimmune indications
Competitive Landscape
- Current Standard: Chronic immunosuppression with corticosteroids, biologics, and cytotoxic agents
- Emerging Alternatives: B cell depletion with anti-CD20 antibodies (rituximab, ocrelizumab)
- Cell Therapy Innovation: First-in-class CAR-NK approach offers potential advantages over antibody-mediated depletion
- Durability Advantage: Single or limited-dose cell therapy versus chronic biologic administration
Development Timeline & Next Steps
- Clinical Stage: First-in-human studies expected to initiate in 2026
- Regulatory Pathway: NMPA approval enables comprehensive clinical development in China
- Manufacturing: Scalable CAR-NK production platform critical for commercial viability
- Global Strategy: Potential for international partnerships following Chinese clinical validation
Forward‑Looking Statements
This brief contains forward-looking statements regarding clinical development, regulatory approvals, and commercial potential for KN5501. Actual results may differ due to risks including clinical trial outcomes, manufacturing challenges, and competitive dynamics.-Fineline Info & Tech