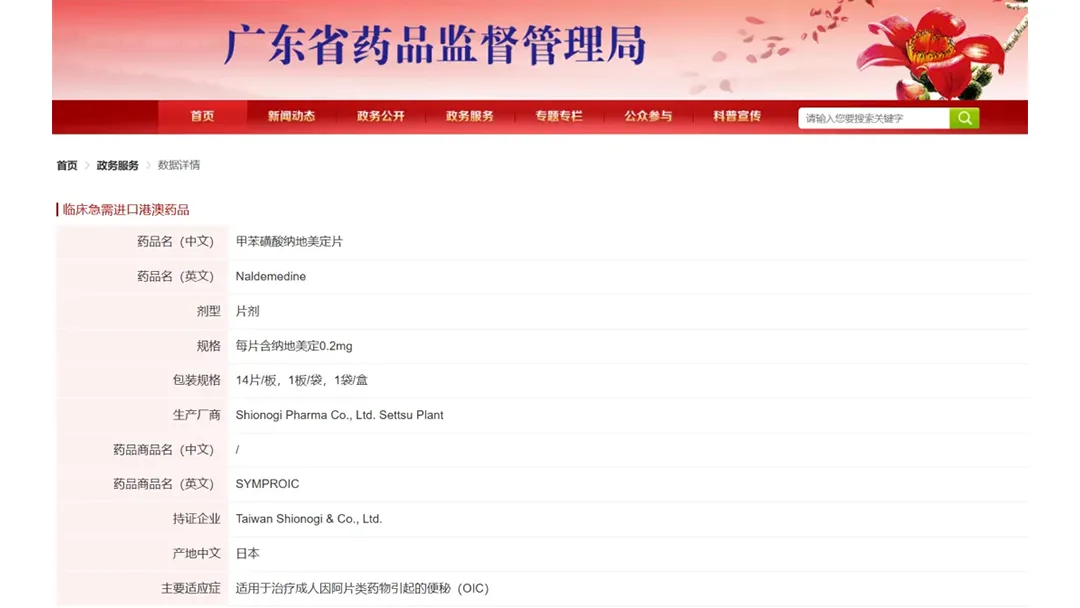
Chia Tai Tianqing Pharmaceutical Group Co., Ltd. (CTTQ), a wholly‑owned subsidiary of Sino Biopharmaceutical Limited (HKG: 1177), announced that the Guangdong Provincial Medical Products Administration (GDMPA) has granted marketing approval for naldemedine, an opioid‑induced constipation (OIC) therapy, for commercialization in the Greater Bay Area (GBA).
Regulatory Milestone
| Item | Detail |
|---|---|
| Agency | GDMPA (Guangdong Province, China) |
| Approval Type | Marketing authorization for GBA region |
| Product | Naldemedine (oral tablets) |
| Indication | Opioid‑induced constipation (OIC) |
| Approval Date | April 2026 |
| Next Steps | Pending NMPA national approval; Hainan Boao pilot program ongoing |
Drug Profile & Mechanism of Action
- Molecule: World’s first approved oral selective peripheral μ‑opioid receptor antagonist (PAMORA)
- Originator: Shionogi & Co., Ltd. (Japan)
- Mechanism: Blocks opioid binding to μ‑receptors in the gut (peripheral action only, no CNS penetration), directly restoring intestinal peristalsis and fluid secretion
- Clinical Advantage: Reverses OIC at its source without compromising central analgesic effects of opioids
- Global Status: Registered in US, EU, Japan, Hong Kong, Macau, and Taiwan; recommended by AGA and ESMO guidelines
Strategic Licensing & Market Entry
| Milestone | Date | Detail |
|---|---|---|
| Exclusive License Secured | Jan 2025 | CTTQ obtains mainland China rights from Shionogi |
| Hainan Boao Pilot | 2025‑2026 | Introduced as clinically urgent‑need drug under special policy zone |
| GDMPA Approval | Apr 2026 | GBA commercialization green‑lit |
| NMPA Review | Ongoing | National marketing application under regulatory review |
Market Impact & Outlook
- China OIC Landscape: With rising opioid prescriptions for cancer pain and post‑surgical care, OIC affects an estimated 15‑20 million chronic opioid users; current treatment options remain limited.
- GBA Advantage: The Greater Bay Area (pop. ~86 million) serves as a high‑value entry point with premium healthcare infrastructure and cross‑border patient flow from Hong Kong/Macao.
- Revenue Forecast: Sino Biopharm projects ¥800 million–¥1.2 billion (US$110 million–US$165 million) in peak annual sales by 2028, assuming successful NMPA approval and tier‑1/2 city rollout.
- Competitive Edge: As the first‑in‑class oral PAMORA, naldemedine holds category leadership over traditional laxatives and competing injectable therapies; guideline endorsements support rapid formulary adoption.
- Regulatory Pathway: GDMPA approval leverages GBA’s accelerated review mechanism for globally validated drugs; NMPA decision expected within 12–18 months, potentially enabling national launch by late 2027.
Forward‑Looking Statements
This brief contains forward‑looking statements regarding regulatory timelines, commercial projections, and market penetration for naldemedine. Actual results may differ due to risks including NMPA approval delays, reimbursement negotiations, competitive entry, and prescribing pattern variations.-Fineline Info & Tech