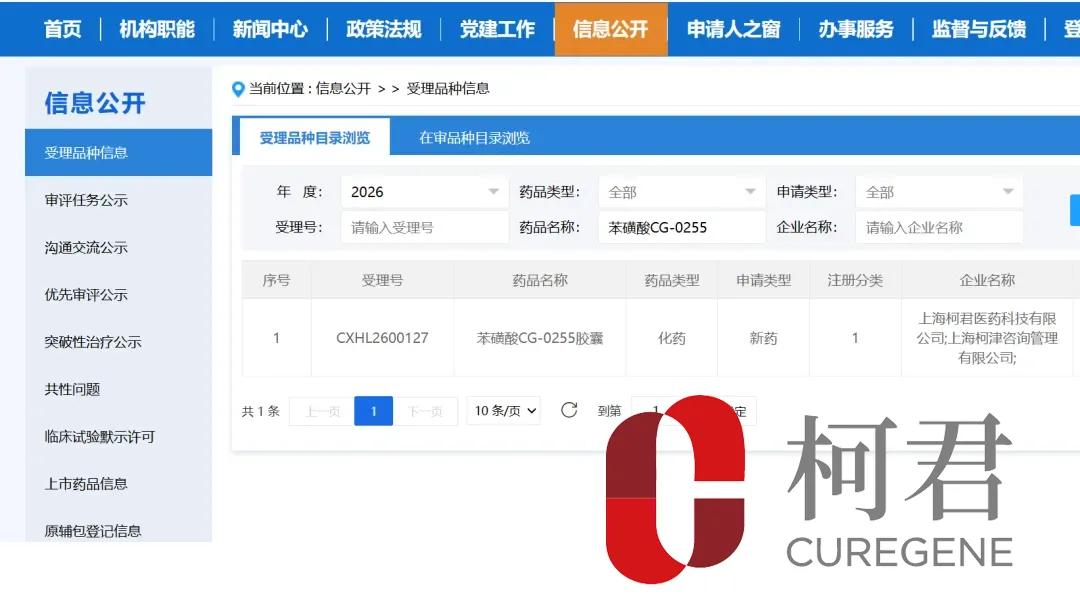
Shanghai CureGene Pharmaceutical Co., Ltd announced it has received clinical trial approval from China’s National Medical Products Administration (NMPA) to initiate a study of its Category 1 innovative drug CG-0255 for the treatment of ischemic stroke. The next-generation antiplatelet agent represents the world’s first P2Y12 receptor inhibitor based on a thiol-based prodrug design, available in both intravenous and oral formulations.
Regulatory Milestone
| Parameter | Details |
|---|---|
| Company | Shanghai CureGene Pharmaceutical Co., Ltd |
| Regulatory Authority | National Medical Products Administration (NMPA) |
| Drug Classification | Category 1 innovative drug |
| Indication | Ischemic stroke |
| Development Stage | Clinical trial approval granted |
| Molecular Innovation | First thiol-based P2Y12 receptor inhibitor globally |
Drug Profile & Technological Innovation
- Mechanism: P2Y12 receptor inhibition with novel thiol-based prodrug design
- Dual Formulations: Available as both intravenous (acute setting) and oral (maintenance therapy) preparations
- Metabolic Advantage: Single-step hydrolysis directly releases active metabolite, bypassing CYP2C19 metabolic pathway
- Clopidogrel Resistance Solution: Fundamentally overcomes “clopidogrel resistance” caused by CYP2C19 enzyme deficiency affecting 30-50% of Asian populations
- Key Advantages:
- Rapid onset of action
- Strong antiplatelet efficacy
- Low inter-patient variability
- Minimal drug-drug interaction risk
Clinical Significance
Addressing Unmet Need
- Ischemic Stroke Burden: China experiences approximately 2.5 million new ischemic stroke cases annually
- Current Limitations: Standard antiplatelet therapies show variable response rates due to genetic polymorphisms
- CYP2C19 Challenge: High prevalence of loss-of-function alleles in Asian populations reduces clopidogrel effectiveness
- Therapeutic Gap: Need for reliable, predictable antiplatelet therapy in acute stroke management
Competitive Differentiation
- First-Mover Status: World’s first thiol-based P2Y12 inhibitor with dual IV/oral availability
- Pharmacokinetic Superiority: Predictable metabolism independent of genetic variations
- Clinical Flexibility: Seamless transition from IV to oral therapy in hospital-to-home care continuum
- Safety Profile: Reduced bleeding risk through consistent, controlled antiplatelet effect
Strategic Implications
Market Opportunity
- China Antiplatelet Market: Valued at RMB 8 billion ($1.2 billion) annually with premium pricing potential for superior agents
- Global Potential: Technology platform applicable to other indications requiring antiplatelet therapy
- Reimbursement Positioning: Category 1 innovative drug status supports favorable NRDL negotiation position
- International Expansion: Novel mechanism provides foundation for global intellectual property protection
Development Strategy
- Initial Focus: Ischemic stroke indication with high unmet need and clear regulatory pathway
- Pipeline Expansion: Potential evaluation in acute coronary syndrome, peripheral artery disease, and other thrombotic conditions
- Combination Therapy: Opportunities for use with other stroke therapeutics including thrombolytics
- Manufacturing Readiness: Dual formulation development demonstrates advanced pharmaceutical capabilities
Next Steps
CureGene will now initiate patient enrollment in the Phase I/II clinical trial, focusing on safety, tolerability, and preliminary efficacy endpoints. The company plans to leverage China’s expedited review pathways for innovative drugs addressing critical unmet medical needs.
Forward‑Looking Statements
This brief contains forward-looking statements regarding clinical development, regulatory pathways, and commercial expectations for CG-0255. Actual results may differ due to risks inherent in pharmaceutical development including clinical trial outcomes, regulatory approvals, competitive dynamics, and market acceptance.-Fineline Info & Tech