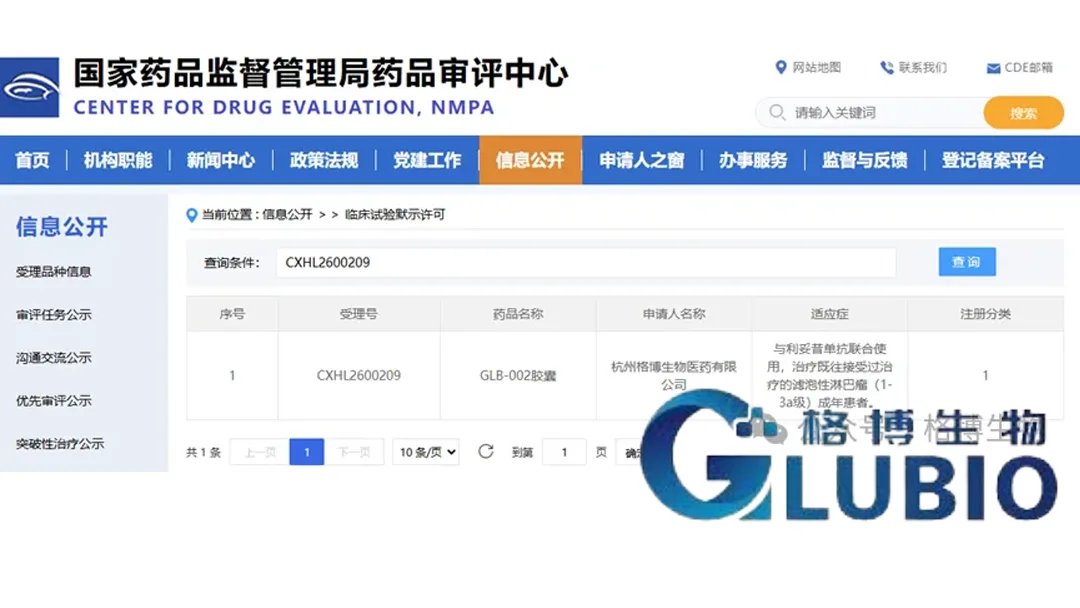
GluBio Pharmaceutical Co., Ltd., a molecular glue targeted protein degradation (TPD) specialist based in Zhejiang, announced it has received clinical trial approval from China’s National Medical Products Administration (NMPA) to initiate a Phase II study of GLB-002 in combination with rituximab for patients with relapsed/refractory follicular lymphoma (R/R FL).
Regulatory Milestone Summary
| Item | Detail |
|---|---|
| Approval Authority | National Medical Products Administration (NMPA), China |
| Development Stage | Phase II clinical trial |
| Product | GLB-002 (selective IKZF1/IKZF3 molecular glue degrader) |
| Combination Partner | Rituximab |
| Target Population | Relapsed/refractory follicular lymphoma (R/R FL) patients |
| Trial Design | Combination therapy assessment |
Technology Profile & Mechanism of Action
- Molecular Class: Next-generation selective IKZF1/IKZF3 molecular glue degrader
- Therapeutic Platform: Targeted protein degradation (TPD) technology
- Mechanism: Induces selective degradation of IKZF1 and IKZF3 transcription factors critical for B-cell malignancy survival
- Innovation: Represents cutting-edge molecular glue approach vs. traditional small molecule inhibitors
- Selectivity: Enhanced specificity for lymphoma-relevant targets compared to first-generation degraders
Clinical Evidence from Phase I
| Patient Population | Key Findings |
|---|---|
| Relapsed/Refractory Non-Hodgkin Lymphoma (R/R NHL) | High overall response rate and durable remission duration |
| Follicular Lymphoma Subgroup | Particularly robust efficacy signals |
| Marginal Zone Lymphoma Subgroup | Strong therapeutic activity observed |
| Safety Profile | Favorable tolerability with manageable adverse events |
The Phase I data demonstrated GLB-002’s potential to address significant unmet needs in indolent B-cell lymphomas where current therapies often fail to provide durable responses.
Market Context & Therapeutic Opportunity
- Follicular Lymphoma Burden: Accounts for approximately 20–30% of all non-Hodgkin lymphomas globally
- Unmet Medical Need: Limited effective options for relapsed/refractory patients after standard therapies fail
- Competitive Landscape: Molecular glue degraders represent emerging class with potential advantages over PROTACs and traditional inhibitors
- Global Potential: Success in China could accelerate international development given strong preclinical rationale
Strategic Implications
- Chinese TPD Leadership: Positions GluBio as pioneer in molecular glue therapeutics among Chinese biotech companies
- Platform Validation: Successful advancement validates GluBio’s proprietary TPD discovery platform
- Partnership Potential: Phase II data could attract global pharmaceutical partnerships for international development
- Pipeline Expansion: Demonstrates feasibility of molecular glue approach for other hematologic malignancies
Forward-Looking Statements
This brief contains forward-looking statements regarding clinical development, regulatory approvals, and therapeutic potential. Actual results may differ due to risks including clinical trial outcomes, safety findings, and competitive developments in the targeted protein degradation field.-Fineline Info & Tech