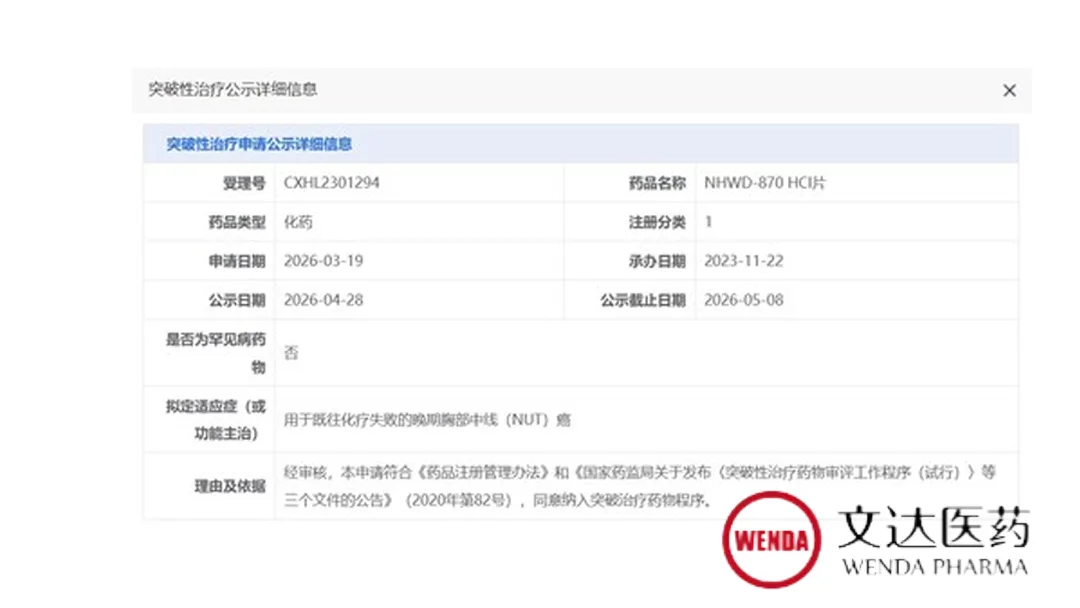
Zhejiang Wenda Pharmaceutical Technology Co., Ltd. announced it has received Breakthrough Therapy Designation (BTD) from China’s National Medical Products Administration (NMPA) for NHWD-870 HCI, its oral BET inhibitor, for the treatment of advanced thoracic midline (NUT) carcinoma in patients who have failed prior chemotherapy. This marks a critical advancement for a devastating rare cancer with no currently approved targeted therapies worldwide.
Disease Burden & Unmet Need
| Parameter | Detail |
|---|---|
| Disease | NUT carcinoma (NUT midline carcinoma) |
| Genetic Marker | NUTM1 gene rearrangement |
| Median Overall Survival | 6.5 months (historical standard) |
| Typical Onset Age | 23.6 years (median) |
| Common Locations | Lungs and head-and-neck regions |
| Diagnostic Challenges | High rates of misdiagnosis and underdiagnosis due to non-specific symptoms |
| Current Treatment Landscape | No effective targeted therapies available globally |
NUT carcinoma represents one of oncology’s most aggressive and challenging malignancies, typically affecting young adults and progressing rapidly to advanced stages before diagnosis.
Clinical Data Supporting BTD
The Breakthrough Therapy Designation is based on compelling efficacy data from Wenda’s Phase II clinical study:
| Endpoint | Result |
|---|---|
| Study Population | 40 evaluable patients with advanced NUT carcinoma (as of December 27, 2025) |
| Objective Response Rate (ORR) | 45.00% in 20 patients with advanced thoracic NUT carcinoma |
| Median Overall Survival (mOS) | 9.33 months for thoracic NUT carcinoma patients |
| Overall Study mOS | 9.33 months (entire population) |
| Survival Benefit | Significant extension compared to conventional chemotherapy (6.5 months historical) |
| Safety Profile | Generally well-tolerated with high potential for clinical application |
The 45% ORR and 9.33-month median survival represent substantial improvements over the natural history of this aggressive disease.
Drug Profile & Mechanism
- Compound: NHWD-870 HCI
- Class: Oral BET (bromodomain and extra-terminal) inhibitor
- Administration: Oral formulation enabling convenient outpatient treatment
- Target Rationale: BET proteins play critical roles in oncogenic transcription programs driven by NUTM1 rearrangements
- Development Stage: Phase II clinical trials with BTD status accelerating potential approval pathway
Strategic & Market Context
- Orphan Drug Opportunity: Ultra-rare disease designation supports premium pricing and expedited regulatory pathways
- Global First-Mover Potential: No approved targeted therapies exist worldwide, positioning NHWD-870 as potentially the first approved treatment
- Young Patient Population: Median onset age of 23.6 years creates significant quality-of-life and survival impact potential
- Diagnostic Awareness: BTD may drive increased diagnostic testing and awareness among oncologists
- Platform Validation: Success validates Wenda’s BET inhibitor platform for other transcription-driven cancers
This BTD represents a beacon of hope for NUT carcinoma patients and their families, addressing one of the most urgent unmet needs in rare oncology.
Forward‑Looking Statements
This brief contains forward-looking statements regarding clinical development, regulatory approvals, and therapeutic potential. Actual results may differ due to risks including clinical trial outcomes, safety findings, regulatory decisions, and diagnostic adoption rates.-Fineline Info & Tech